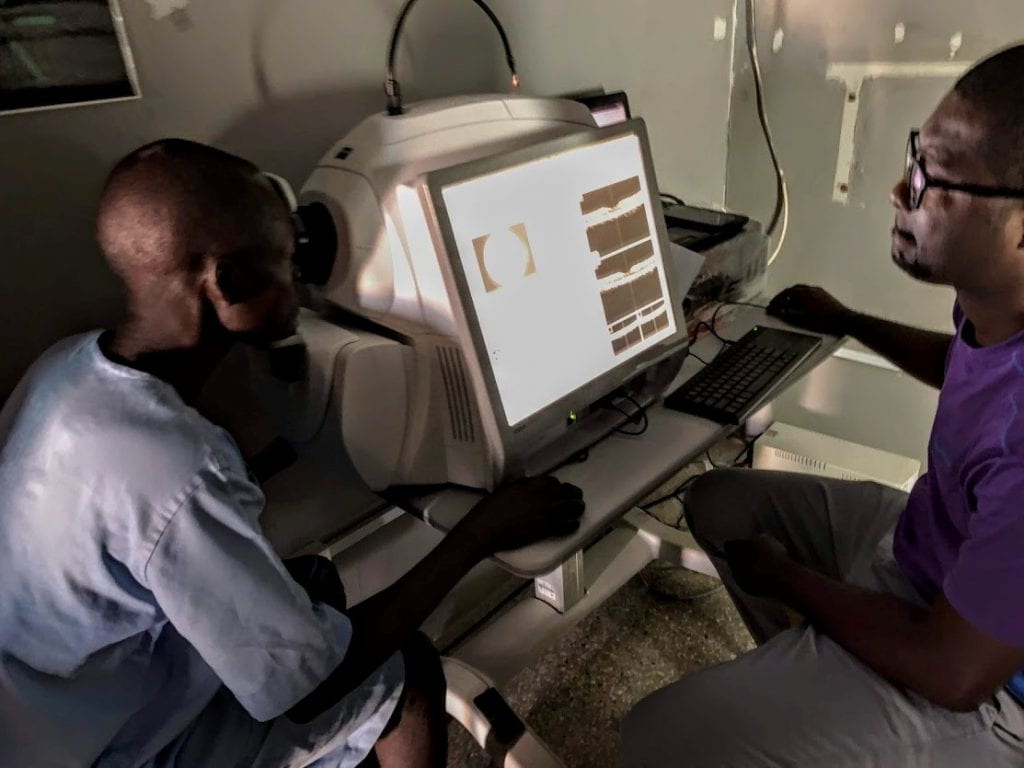
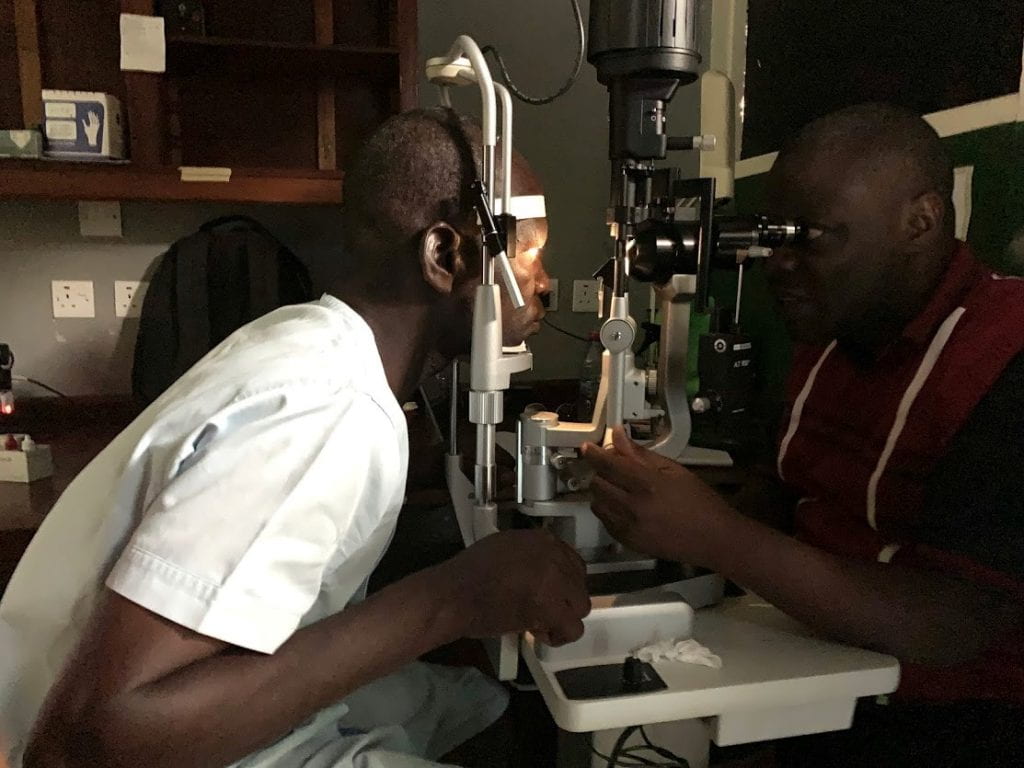
DOLF researchers and staff traveled to Hohoe, Ghana in December 2019 to work with Nick Opoku’s team at the University of Health and Allied Sciences to initiate a trial of the triple drug treatment IDA in persons with onchocerciasis. This study is testing the same IDA combination (ivermectin plus DEC and albendazole) that is so effective for clearing microfilariae from the blood of persons with lymphatic filariasis. Participants in the oncho/IDA study were pre-treated with ivermectin months before IDA treatment to clear microfilariae from the skin and eyes to reduce the risk of adverse events following IDA treatment. Study procedures include extensive ophthalmological testing with slit lamp examinations, retinal photography, and ocular coherence tomography (OCT) before and after treatment to detect and document ocular adverse events. In addition to safety assessments, the study will compare the effects of IDA to the dual drug regimen ivermectin plus albendazole for suppressing the reappearance of Mf in the skin and for killing or sterilizing adult Onchocerca volvulus worms.